|
The principal quantum number equals the number of sublevels within that principal energy level. The higher the energy level occupied by an electron the more the energetic it is. The maximum number of electrons that can occupy the fourth principal energy of an atom is 32. The number of sublevels in an energy level is equal to the square of the principal quantum number of that energy level. electron configuration The most stable arrangement of electrons around the nucleus of an atom. neon 1s^2,2s^2,2p^6 Aufbau principle Electrons enter orbitals of lowest energy first. Pauli exclusion principle An atomic orbital may describe at most two electrons. Hunds rule When electrons contain orbitals of equal energy, one electron enters each orbital until all the orbitals contain one electron with parallel spins. The Pauli exclusion principle states that an atomic orbital may describe at most two electrons. false T/FĪs many as four electrons can occupy the same orbital True T/F 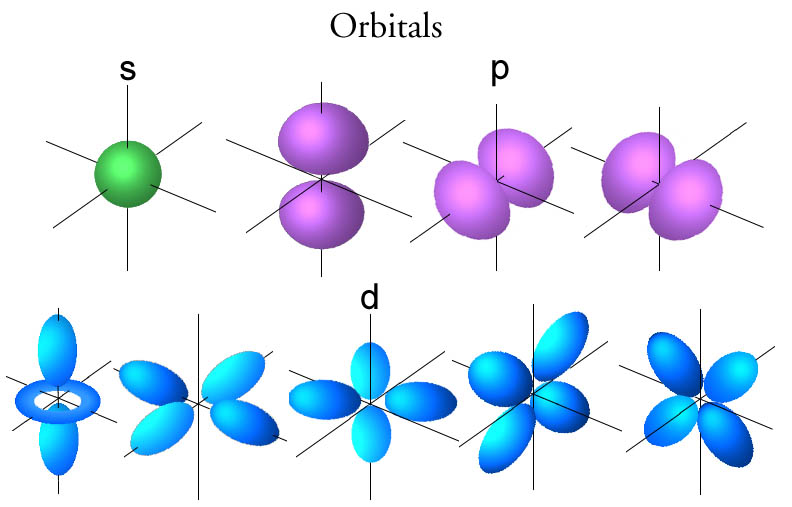
The configuration 3d^4,4s^2 is more stable than the configuration 3d^5,4s^1. The orbitals of a principal energy level are lower in energy than the orbitals in the next higher principal energy level. _ and copper are exceptions to the Aufbau principle. chronium Correct electron configurations can be obtained by using the Aufbau diagram for the elements up to and including vanadium. electrons When using the shorthand method for showing the electron configuration of and atom, superscripts are used to indicate the number of _ occupying each sublevel. superscripts When using the shorthand method for showing the electron configuration of an atom, _ are used to indicate the number of electrons occupying each sublevel. an electron Hund's rule states that the electrons pair up after each orbital in a sublevel is occupied by _. opposite To occupy the same orbital, two electrons must have _ spins. 2 The Pauli exclusion principle states that a maximum of only _ electrons can occupy each orbital. Pauli exclusion The _ principle states that a maximum of only 2 electrons can occupy each orbital. consistent The various orbitals within a sublevel of a principle energy level are always of _ energy. Aufbau principle The _ describes the sequence in which orbitals are filled. electron configurations The ways in which electrons are arranged around the nuclei of atoms are called _. The electron probability clouds for atomic orbitals are spherical in shape. true T/FĮlectrons must have a certain minimum amount of energy called a quantum in order to move from one energy level to the next higher energy level. probability This model estimates the _ of finding an electron within a certain volume of space surrounding the nucleus. Quantum mechanic The _ model is the modern description of the electrons in atoms. circular In the Bohr model, the electrons move in _ paths. nucleus Rutherford pictured the atom as a dense_ surrounded by electrons. Thomson's model is referred to as the _ model. Thompson After the discovery of the electron by _, the atomic model was revised to include them. Dalton The first modern atomic theory, proposed by _ portrayed the atom as a solid, indivisible mass. 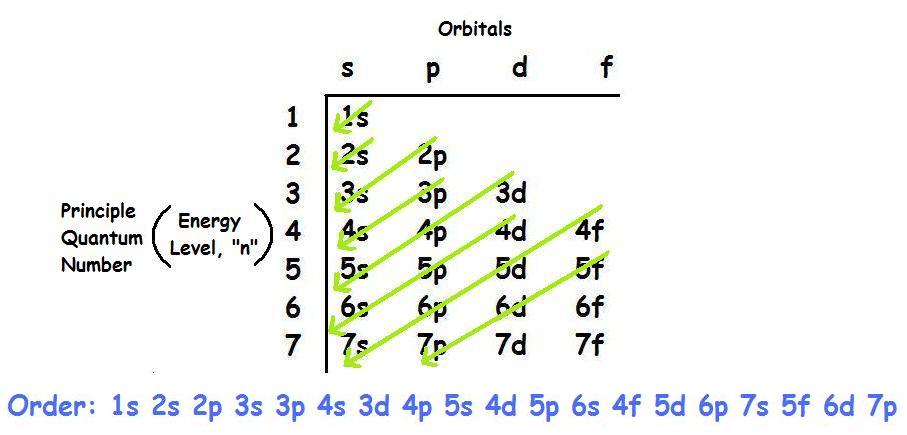
electrons The chemical properties of atoms, ions, and molecules are related to the arrangement of the _ within them. Aufbau principle Electron configurations can be written using the _, the Pauli exclusion principle, and Hundi's rule. atomic orbitals Orbitals, which are found in _, have different shapes, depending on the energy of the electrons they contain. quantum mechanical The _ model describes the energy an electron can have and the probability of finding the electron in various locations around the nucleus of an atom. orbitals Bohr proposed that and electron is found only in specific circular paths, or _, around the nucleus. stable Half-filled levels are not as_ as filled levels but are more stable than other configurations. stable Filled energy sublevels are more _ than partially filled sublevels. the number of electrons occupying each sublevel In an electron configuration, what does a superscript stand for? number of electrons in the atom In an electron configuration, what does the sum of the superscripts equal? false T/FĮvery element in the periodic table follows the aufbau principle. Hund's Rule When electrons occupy orbitals of equal energy, one electron enters each orbital until all the orbitals contain one electron with the same spin direction. Pauli exclusion principle Electrons occupy orbitals of lowest energy first. electron configuration The ways which electrons are arranged into orbitals around the nuclei of atoms are called_ pauli exclusion principle An atomic orbital may describe at most two electrons. Principle quantum numbers (n) the term that is used to label the energy levels of electrons S The letter _ is used to denote a spherical orbital.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
